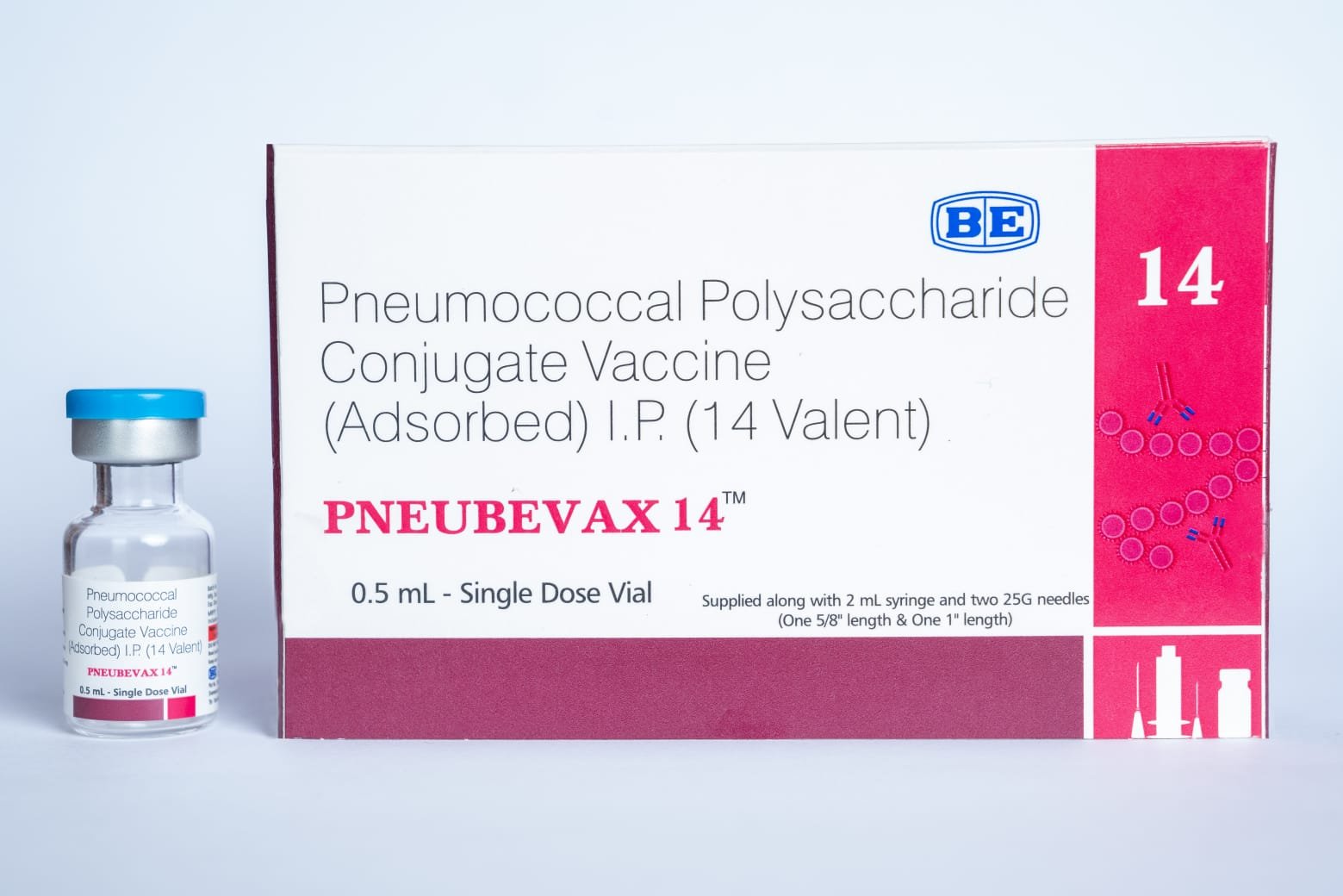
Biological E’s PNEUBEVAX 14® vaccine receives WHO’s pre-qualification
November 20, 2025 | Thursday | News
PNEUBEVAX 14® is Biological E. Limited’s 14-valent Pneumococcal Conjugate Vaccine
Biological E. Limited (BE), a leading Hyderabad-based vaccine and pharmaceutical company, has announced that the World Health Organisation (WHO) has granted Pre-qualification (PQ) status to their 14-valent Pneumococcal Conjugate Vaccine (PCV), PNEUBEVAX 14® (BE-PCV-14).
PNEUBEVAX 14® becomes BE’s 11th WHO pre-qualified vaccine. Developed specifically to prevent invasive pneumococcal diseases, PNEUBEVAX 14® protects against infections caused by 14 different Streptococcus pneumoniae serotypes, including serotypes 22F and 33F—two strains not covered by select other PCVs.
Administered to infants from six weeks of age, PNEUBEVAX 14® is designed to guard against serious diseases such as pneumonia, meningitis, and sepsis as part of the primary vaccination schedule.
Commenting on the WHO pre-qualification, Mahima Datla, Managing Director, Biological E. Limited, said, “This milestone enhances global access to high-quality pneumococcal vaccines and strengthens supply security, particularly for children who need them most. WHO PQ enables broader reach through global immunisation initiatives, and we remain committed to supporting public health partners in delivering affordable, reliable vaccines across India and around the world.”