How Early Diagnostic Tech Can Stop Cancer In Its Tracks
February 04, 2023 | Saturday | Views | By Suraj Nair, Lead (TechSprouts), Ankur Capital
Surviving any form of cancer is challenging. It is more challenging if detected in the later stages. Data suggests that average 5-year survival of patients who were detected with cancer at an early stage was 91 per cent which dropped to 26 per cent at late stages. Nearly 10 million deaths were reported in 2020 due to cancer, almost half of which were from breast and lung cancer put together. Detection of cancers at early stages is therefore very important for effective therapy.

Traditionally, there were two broad techniques available for early screening and diagnosis of cancers: tissue biopsy and the use of radiographic imaging. Tissue biopsy involves observing under the microscope, a small tissue extracted from the area of the body which may be cancerous. This is performed by a pathologist. In most cases, this is done only after symptoms are seen and is therefore used for diagnosis rather than screening. Tissue biopsy, however has several drawbacks- it is invasive in nature and therefore painful, its costly and time consuming and struggles to capture the heterogeneity of the tumors effectively. Therefore, it is not an effective strategy for early detection of cancers.
Digital pathology emerged as a tool to reduce the burden on pathologists and avoid manual errors in analysing the images. Digital images of tissue biopsies can be stored on secure servers and subsequently analysed using Artificial Intelligence (AI) tools.
Ibex Medical Analytics is one of the pioneers in the field of using AI in biopsies and has raised more than $50 million in venture capital funding. Paige and PathAI are just some of the other companies in this space. Despite its immense potential, penetration of digital pathology tools in the clinical settings has remained low due to challenges in digitising 100 per cent of tissue biopsy images and an inherent inertia in the system to adopt digitisation. The growth of the digital pathology market will depend heavily on the ability of the early adopters to scale up the technology into larger clinical settings and set examples for the late movers in the market.
Another technique which has emerged since the early 2000s is the liquid biopsy test. This test involves analysis of cancer tumor cells (CTC) or other cancer biomarkers such as circulating tumor DNA (ctDNA), exosomes and miRNAs in blood, plasma, serum, saliva or urine samples. This provides information on the genomic and proteomic profile of the cancer cells and can help in effective precision medicine to be used in therapy. One of the major advantages of a liquid biopsy test is that it is non-invasive, which means it reduces pain, injury or infections which are often associated with tissue biopsies. Moreover, serial liquid biopsy can help in detecting the heterogeneities in tumors and their genetic profile. Liquid biopsy can be used for screening and diagnosis of cancers. The first clinically approved liquid biopsy test was commercialised in 2004 by Menarini Silicon Biosystems to detect advanced breast cancer, colorectal cancer, and prostate cancer. However, liquid biopsy based on ctDNA analysis was only accepted as a diagnostic tool in the US in 2016. Since then, a few other tests have become commercially available-
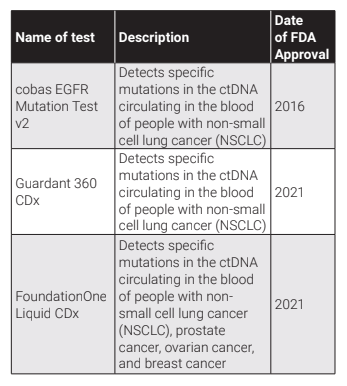
The use of liquid biopsy as a tool for cancer screening and diagnosis is still in its nascency and more trials and validations need to be conducted for the products to be widely accepted in the market. However, a plethora of genetic and proteomic information on humans has been generated over the last decade which has encouraged a multi-omics approach to the discovery of cancer biomarkers. This is expected to open up opportunities to detect tumors which currently have no early diagnosis, track the progress of cancers during therapy and identify very specific mutations regularly. Moreover, use of advanced AI tools trained on the multi-omics information can reduce the timelines for identifying unique biomarkers. Finally, screening of multiple cancers from a single test is an avenue which has seen major developments.
The field has seen significant venture capital investments in the last few years. 2021 saw close to $800 million invested into companies building cancer screening tools. Delfi Diagnostics, a company developing AI based liquid biopsy tests raised a series B round of $225 million in 2021 for a total funding of $330 million. The company is developing single and multi-cancer liquid biopsy blood tests. The company is currently validating the technology for early detection of lung and other cancers in the CASCADE-LUNG clinical trial.
Grail, which is developing a multi-cancer blood based detection test, raised around $390 million in 2020, for a total funding of more than $1.9 billion. The company is currently collecting clinical data on over 73,000 participants through two clinical trials. Similarly, Thrive Early Detection raised a $257 million series B to commercialise its multi-cancer blood test.
Many other companies in this space such as Freenome, Epic Sciences, Guardant Health, Nucleix, Inivata, NAVERIS, Genecast, AccuraGen, Cirina, Jiyinga Technology, Resolution Bioscience, Haystack Oncology have raised significant private funding over the last few years.
One of the most interesting companies in the AI personal medicine market is Imagene AI, which is using deep learning algorithms to reduce the time for identification of biomarkers from many weeks to just under 2 minutes. The company has raised more than $21 million.
In India, Strand Life Sciences launched its liquid biopsy tests for detecting lung cancer, colorectal cancer, breast cancer and bladder cancer in 2018. Actorius Innovations and Research has also developed a liquid biopsy test for lung, breast, colorectal, head and neck cancers. Their technology has already been approved by the Central Drugs Standard Control Organisation (CDSCO). Oncophenomics is another company which is commercialising a liquid biopsy test in India.
Radiographic imaging, on the other hand, has been recommended only for three types of cancers as of today- breast cancer, colorectal cancer and lung cancer. Imaging is however also done for screening of other cancers such as colon, kidneys, cervix among others. Imaging techniques are used primarily for screening of these cancers and are usually followed by a biopsy test for diagnosis. However, newer technologies are also helping in accurate diagnosis. Scientists and pathologists continue to debate on the risk to reward benefits of the various imaging modalities- radiology imaging may even harm the healthy cells and tissues due to prolonged exposure times, thereby leaving them at risk of developing cancer later.
To overcome these challenges, new technologies are emerging along with the use of AI/machine learning (ML) tools to enable faster and accurate screening of cancers. While traditional methods including PET, X-ray, CT-Scans, MRI, Mammography and ultrasound have seen gradual improvements over the years, newer tools with the ability to identify patterns in images using algorithms have been able to disrupt the industry and lower the TAT for the screening of cancers. Apart from the larger companies in this market such as Siemens Healthineers, startups such as Niramai, Kheiron Medical, Deepwise, SkinVision and Lunit are also making their mark.
Niramai has built an alternative to mammography by using thermal imaging along with an AI/ML software for early detection of breast cancers. Mammography, which is recommended to be done every two years for women between the age group of 50 and 74, is painful and highly uncomfortable for women. Niramai’s technology on the other hand, is highly non-invasive, radiation free and can be performed without any pain and discomfort. It is also recommended for women of all ages. Its patented machine learning algorithm analyses the images obtained and is able to accurately detect the occurrence of breast cancer at a very early stage. Their medical device recently received US FDA clearance. The company has raised more than $6 million in private funding.
Kheiron Medical has also built a deep learning AI platform to assist radiologists in accurate and faster diagnosis of breast cancer. The company has raised more than $22 million in private funding. Many of the tasks in the radiology labs such as screening of images, tumour measurements and segmentation are repetitive and tedious. They are also affected by human errors and incorrect diagnosis. The use of these AI/ML tools can significantly reduce the errors and improve the diagnostic performance.
All said and done, early diagnosis of cancers has seen technological disruption over the last decade, both in biopsies as well as radiography imaging. Large datasets from genomics, proteomics, biopsy images have incentivised the use of artificial intelligence and machine learning tools to process this data and provide accurate and faster diagnosis. Genetic testing for screening these cancers is also picking up.
The next decade will be challenging- these technologies need to scale up and have widespread adoption which will require validation through large heterogeneous datasets. This is especially challenging for liquid biopsies, since identification of patient cohorts comes with biases and the tests currently lack accuracy. Secondly, the inherent inertia in the system will have to be overcome, either by incentivising the use of these new technologies through insurance payer models or by enabling policies mandating their use as part of the patient journey. Last, but not the least, patients will have to be educated on the advantages of using these tools for cancer screening and diagnosis.
Suraj Nair, Lead (TechSprouts), Ankur Capital











